Aran Biomedical Attending EHS 2019
Aran Biomedical will be attending the European Hernia Society’s annual congress. The event takes place in Hamburg, from the 11th – 14th of September 2019.
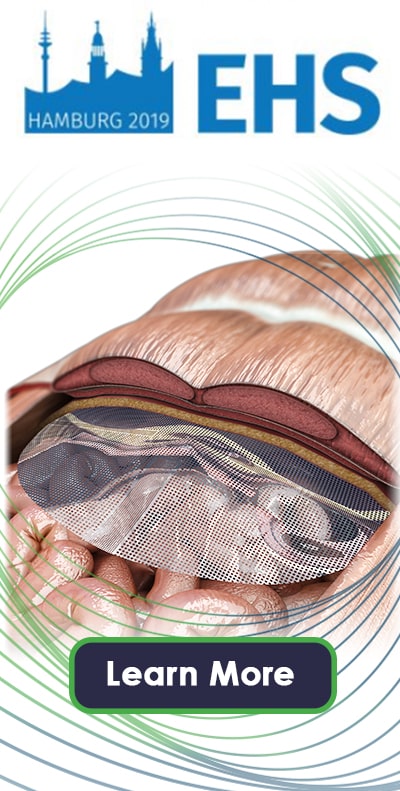
Aran is an OEM Supplier and trusted partner in the General Surgery market, leveraging its history as Legal Manufacturer of a portfolio of hernia mesh products (MotifMESH, VitaMESH & VitaMESH Blue). We offer end-to-end mesh development services, from Design Input at concept phase to the supply of finished Commercial Product. Offerings include condensed flat-sheet mesh tailored to minimally-invasive repair, composite mesh solutions, and long-term absorbable mesh.
Representatives from Aran will be available to discus your product requirements.
To schedule a meeting during the show, please contact info@aranbiomedical.com
Implantable Medical Textiles
- Aran Biomedical offer a full suite of textile development capabilities, including finishing, assembly & packaging solutions. We can develop knitted, woven & braided constructs, in flat & tubular structures.
Absorbable Implants
- The company has extensive knowledge of resorbable implants, and significant experience working with resorbable polymers. Product solutions include fully-absorbable mesh, semi-resorbable mesh, and resorbable barrier layer mesh.
ProTEX MED – Implantable Grade Polypropylene Mesh
- ProTEX Med™, is a custom synthesised PP resin, exclusively available for partners leveraging downstream textile conversion processes. It is certified to ISO 10993-5 and comes with a unique FDA Device Master File reference, available to resin users to support associated product qualification.










Leave A Comment
You must be logged in to post a comment.